|
∆H sol = -80 KJ/mol Enthalpy Change of Hydration It is the amount of heat absorbed or liberated when a substance is dissolved in a solvent to form an infinitely dilute solution.Įnthalpy change of solution may be positive or negative and is denoted by ∆H sol. The value of enthalpy change is positive because this reaction is endothermic. It is the enthalpy change when one mole of gaseous atom(s) is formed from the element. How to Find Delta H? Enthalpy Change of Atomisation.These enthalpy changes are of several types. The absolute value of enthalpy cannot be determined but we can measure the change in enthalpy. It is the sum of internal energy and pressure-volume work. If the energy of a system decreases, the surrounding energy increases, but the total energy remains constant and vice versa. So, we can say that the energy of the universe is constant. It can also be explained as the total energy contained inside a system.Īccording to the first law of thermodynamics, energy can neither be created nor be destroyed, it remains constant. There are some similarities between enthalpy and entropy as well which are such that, both enthalpy and entropy areĮnthalpy is the amount of energy either lost or gained by the system. According to the 2nd law of thermodynamics, the entropy of the According to the 1st law of thermodynamics, the energy of the Entropy is negative for non-spontaneous processes. Enthalpy is negative for exothermic processes. 
Entropy is positive for spontaneous processes. Enthalpy is positive for endothermic processes. Entropy change in a cyclic process is zero. Enthalpy change in a cyclic process is zero. In a spontaneous process, change in entropy will not be less in the direction of spontaneity. Change in enthalpy is equal to the energy supplied as heat at constant pressure. H = U+PV dS = d/T Its unit is KiloJoules/mole. It is the amount of heat transferred reversibly in and out of the system at a given temperature. It is the sum of internal energy and product of PV work. It is the measurement of the disorderness of a system. This lost energy is called the ’entropy’.Įnthalpy Entropy It is a measurement of energy. The concept of entropy develops from the fact that for energy to be converted to work, some dissipations must occur. This idea comes from the concept that heat always flows from hot to cold regions spontaneously which is equal to the entropy change. The term entropy was introduced by the scientist Rudolf Clausius in 1850. It is an important quantity because most of the reactions occur at constant pressure and it is used to measure the heat of a reaction. Enthalpy tells us how much heat is added or removed from the system. The word enthalpy means ‘total heat content’. 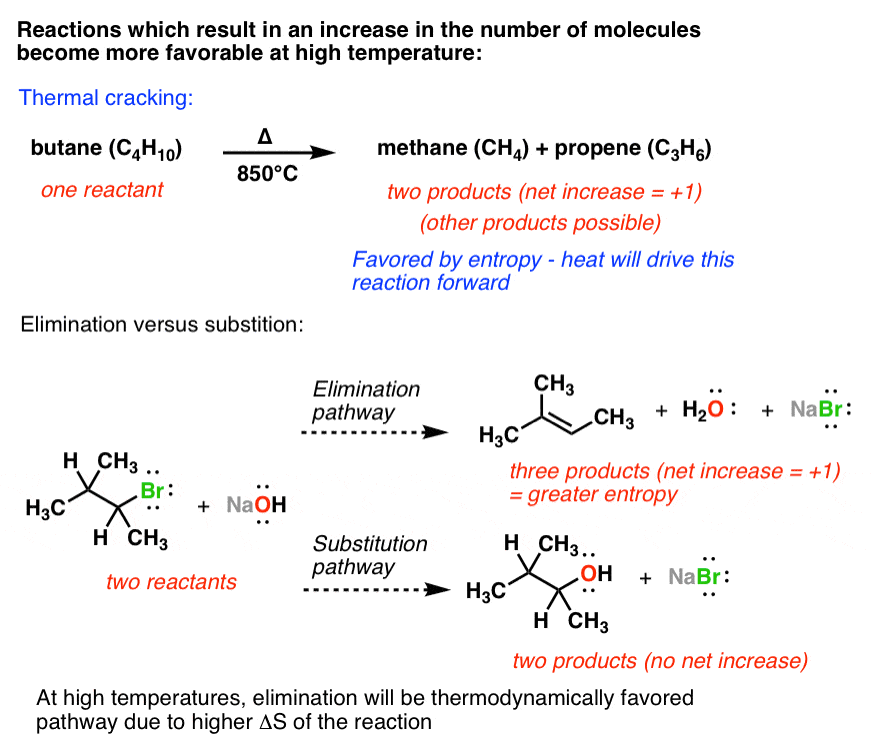
The term enthalpy was introduced by a Dutch Scientist, Heike Kamerlingh Onnes in 1909. In thermodynamics, the change in enthalpy and entropy can be measured rather than their absolute values. 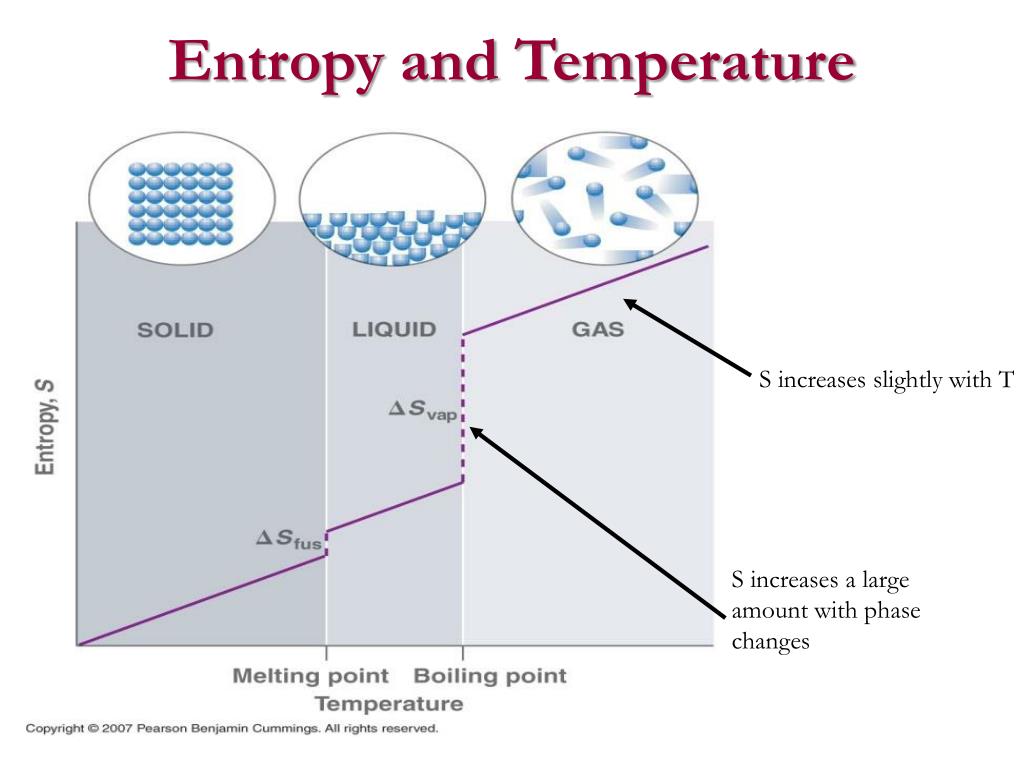
Enthalpy (H) of a thermodynamic system is an energy-like state function property that is equal to the total internal energy (U) and pressure-volume (PV) work whereas entropy is an intrinsic disorderness of a system under certain conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
